Descripción
Selank Peptide Vial
| CAS Number | 129954-34-3 |
| Other Names | Selanc, TP-7, Thr-Lys-Pro-Arg-Pro-Gly-Pro, TP 7, UNII-TS9JR8EP1G, TS9JR8EP1G |
| IUPAC Name | (2S)-1-[2-[[(2S)-1-[(2S)-2-[[(2S)-1-[(2S)-6-amino-2-[[(2S,3R)-2-amino-3-hydroxybutanoyl]amino]hexanoyl]pyrrolidine-2-carbonyl]amino]-5-(diaminomethylideneamino)pentanoyl]pyrrolidine-2-carbonyl]amino]acetyl]pyrrolidine-2-carboxylic acid |
| Molecular Formula | C33H57N11O9 |
| Molecular Weight | 751.9 |
| Purity | ≥99% Pure (LC-MS) |
| Liquid Availability | N/A |
| Powder Availability | N/A |
| Storage Condition | Store cold, keep refrigerated. Do NOT freeze. |
| Terms | All products are for laboratory developmental research USE ONLY. Products are not for human consumption. |
**Información Importante: Cada péptido se entrega liofilizado y debe ser reconstituido con Agua Bacteriostática para poder ser administrado en forma líquida.
Vea aquí el video sobre la reconstitución de péptidos
What is Selank?
Selank is a synthetic peptide derived from the naturally occurring tetrapeptide Tuftsin, known for its immunomodulatory and nootropic properties. The compound has gained attention for its potential to enhance cognitive function, reduce anxiety, and modulate the immune system. Selank is often used in research and clinical settings to investigate its effects on mental health, stress resilience, and immune responses, making it a promising compound in the fields of neuroscience and immunology.
Ver más…
3 Main Research Findings
1) Treatment with Selank was found to prevent formation of ethanol-induced memory and attention disturbances, as well as increases in BDNF levels in the hippocampus and frontal cortex.
2) Administration of Selank was shown to increase the amplitude and discharge rate of spontaneous inhibitory postsynaptic currents in hippocampal pyramidal CA1 neurons.
Selected Data
1) The described experiment conducted by the research team of Kolik et al involved the use of outbred male rats, aged approximately nine months. The rats’ mean body weight ranged between 480 and 510 grams, and they were maintained during the active phase of the experiment. These animals were housed individually in cages within a vivarium environment that adhered to standard conditions, including a 12-hour light/dark cycle, a constant temperature, and regular access to water and pelleted food [1].
To model chronic alcohol intoxication, a group of rats was given 10% ethanol as their sole fluid source. These rats, demonstrating a marked preference for ethanol as determined by an intake of approximately 5.2±0.3 grams of ethanol per kilogram of body weight, exhibited alcohol deprivation effects and were provided with access to both ethanol and water under free-choice conditions. In contrast, an age-matched control group of intact rats, not exposed to ethanol, was also included in the study, serving as a baseline for comparison. The source of ethanol used was Glavspirt, and Selank, a synthetic peptide.
The experimental design involved four groups of rats, which were randomly assigned to ensure unbiased distribution. The first group consisted of intact rats that received daily intraperitoneal injections of water for seven days. The second group comprised intact rats that, similarly, received Selank at a dose of 0.3 mg/kg in a water solution and was administered at 0.1 ml per 100 grams of body weight. The third group included rats subjected to prolonged alcohol exposure for 30 weeks, which subsequently underwent a withdrawal period. These rats received intraperitoneal injections of water for seven days. The fourth group consisted of rats that had experienced the same alcohol exposure and withdrawal, but received Selank injections instead of water during the withdrawal period, at the same dosage as the second group. This grouping allowed for the assessment of Selank’s effects on rats with a history of chronic alcohol intoxication during withdrawal [1].
The cognitive effects of Selank were assessed using the novel object recognition test, a widely used method for evaluating hippocampal memory processes. The test involved several stages including: initial habituation where each rat was introduced to an open field arena with white walls and a 1-2 cm sawdust floor, allowing the animal to acclimate without objects present. After 24 hours, the second stage involved placing two identical 12 cm tall cylindrical plastic boxes, hermetically sealed for food into opposite corners of the arena, 15 centimeters from the walls. During this phase, the total time the rats spent sniffing and exploring each object was recorded, denoted as Tf for familiar objects. In the third stage, one of the familiar objects was replaced by a novel object differing in shape, color, and material, positioned at the same spot as the familiar one. The exploration time of the new object was recorded, labeled Tn. Each stage lasted five minutes, and the discrimination index (DI) was calculated using the formula (Tn – Tf) / (Tn + Tf), providing a measure of the animals’ memory performance characterized by their ability to differentiate novel from familiar objects [1].
On the eighth day after initiating the treatment, corresponding to 24 hours after the final injection of Selank or water, the rats were euthanized by decapitation. Their brains were extracted and dissected on ice, with specific focus on the frontal cortex and hippocampus. These tissues were immediately frozen in liquid nitrogen for preservation. For biochemical analysis, tissue samples were homogenized in a lysis buffer containing 50 mM tris-HCl, 5 mM EDTA, 1 mM DTT, and 1% Triton X-100 at a ratio of 1:10 tissue to buffer, kept at 4°C. The homogenates were then centrifuged at 13,000 rpm for ten minutes at 4°C to separate debris.
To analyze the expression of Brain-Derived Neurotrophic Factor (BDNF) in the brain tissues, proteins from the supernatants were transferred onto PVDF membranes via electroelution for 45 minutes. Western blot analysis was performed by blocking the membranes with TBS-T buffer containing 1% Tween-20 and 5% skimmed milk for one hour, followed by incubation with primary antibodies specific to BDNF at a 1:1000 dilution overnight. After washing, the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies at a 1:2000 dilution for one hour. The detection of BDNF protein was carried out using an ECL reagent system, and the signals were visualized and documented. This method allowed for the quantification of BDNF levels within the frontal cortex and hippocampus, providing insights into the molecular effects of Selank on neurotrophic factors related to memory and neuroplasticity [1].
In summary, this study investigated the impact of Selank on rats with a history of chronic alcohol exposure and withdrawal, focusing on cognitive functions and neurobiological markers. The experimental protocol included control of housing conditions, ethical adherence, and precise behavioral, biochemical, and molecular assessments. The use of the novel object recognition test provided behavioral data related to hippocampal memory performance, while biochemical methods such as Western blotting allowed for the evaluation of neurotrophic factors like BDNF in key brain regions. The findings from this research contribute to understanding the potential neuroprotective and cognitive-modulating effects of Selank, particularly within the context of alcohol-related neurobiological alterations.
2) This study performed by researchers Povarov et al investigated the effects of Selank on spontaneous synaptic activity in rat hippocampal CA1 neurons using cultured hippocampal slices and electrophysiological techniques. The researchers prepared hippocampal slices from the brains of Wistar rats aged 13-19 days, employing a standard technique known to preserve the integrity of neuronal circuits and synaptic connections. After preparation, the slices were incubated for 2 hours at room temperature in a carefully formulated saline solution. This solution contained specific concentrations of essential ions, including 119 mM sodium chloride, 2.5 mM potassium chloride, 1.3 mM magnesium sulfate, 2.5 mM calcium chloride, 1 mM sodium phosphate, 26.2 mM sodium bicarbonate, and 11 mM glucose. The pH of the solution was meticulously adjusted to 7.4 using a mixture of 95% oxygen and 5% carbon dioxide, ensuring a physiological environment conducive to neuronal activity [2].
To isolate and study the electrophysiological properties of individual neurons, the researchers employed a visual identification method using a water immersion objective at ×40 magnification. This allowed them to visualize individual neurons in the slices under transmitted light and select pyramidal neurons in the CA1 region of the hippocampus, the target of their investigation. The electrophysiological parameters of these pyramidal neurons were recorded using a whole-cell voltage clamp technique, a sophisticated method that allows precise control and measurement of membrane potential and current flow. The holding potential, the voltage at which the neuron’s membrane potential was maintained, was set to -70 mV, a typical resting potential for these neurons.
The recording pipette, a fine glass electrode used to establish electrical contact with the neuron, was filled with an internal solution containing specific concentrations of 120 mM potassium chloride, 5 mM sodium chloride, 0.5 mM calcium chloride, 2 mM magnesium chloride, 10 mM EGTA, and 5 mM HEPES, and HEPES-Na. This solution was carefully designed to mimic the intracellular environment of the neuron and to facilitate stable and accurate recordings. The researchers focused on neurons that did not discharge spontaneously, had a resting potential not below -55 mV, and exhibited an access resistance defined by a measure of the electrical resistance between the pipette and the neuron’s interior of ≤30 MΩ. Neurons with access resistance variations exceeding 30% were excluded from the analysis to ensure the quality and reliability of the recordings [2].
The spontaneous postsynaptic currents (PSCs), which represent the electrical activity generated by synaptic transmission, were analyzed off-line using Mini Analysis software. This software allowed the researchers to accurately identify and quantify the peak amplitude and discharge rate of short-term, <300 msec, PSCs. Initially, control recordings were obtained to establish baseline values for peak amplitude and discharge rate before applying Selank. Then, the same recordings were carried out with Selank applied at various concentrations. The experiments were repeated with varying concentrations of Selank to identify and note any changes.
To investigate the source of the observed effects, the researchers performed additional experiments using tetrodotoxin (TTX), a potent neurotoxin that blocks voltage-gated sodium channels and prevents the transmission of action potentials along neuronal axons. By applying TTX, they aimed to determine whether the effects of Selank were due to changes in the activity of interneurons. Finally, the data were analyzed statistically using Wilcoxon’s signed-rank test for paired comparison, a non-parametric test suitable for comparing data from the same neurons before and after treatment [2].
Discussion
1) This research performed by Kolik et al delves into the potential therapeutic benefits of Selank, a peptide analogue of tuftsin, in counteracting the memory-impairing effects of chronic alcohol consumption. The study focuses on the role of BDNF, a crucial protein involved in neuronal development, synaptic plasticity, and cognitive function, in mediating the effects of Selank. The central hypothesis is that Selank can protect against ethanol-induced memory deficits by modulating BDNF levels in key brain regions, specifically the hippocampus and prefrontal cortex, which are known to be vulnerable to alcohol’s toxic effects [1].
The researchers employed an experimental model involving outbred male rats subjected to chronic alcohol intoxication. The rats were provided with a 10% ethanol solution as their sole source of fluid for an extended period of 30 weeks, mimicking the long-term alcohol exposure characteristic of chronic alcoholism. This prolonged ethanol consumption aimed to induce significant memory impairments and alterations in BDNF expression. To assess the potential protective effects of Selank, a group of alcohol-exposed rats received intraperitoneal injections of Selank during alcohol withdrawal, while control groups received either water or were not exposed to alcohol at all. This allowed for a comparative analysis of cognitive function and BDNF levels across different treatment conditions [1].
Cognitive function was evaluated using the novel object recognition test, a widely used behavioral paradigm that assesses short-term memory and object discrimination abilities in rodents. This test relies on the natural tendency of rats to explore novel objects more than familiar ones. By measuring the time spent exploring a novel object compared to a familiar one, researchers could quantify the extent of memory impairment induced by alcohol and the potential cognitive-enhancing effects of Selank.
The study’s findings revealed several key insights into the relationship between chronic alcohol exposure, BDNF, and cognitive function. As expected, chronic alcohol consumption significantly reduced the discrimination index in the novel object recognition test, indicating a marked impairment in short-term memory. This finding underscores the detrimental effects of prolonged alcohol exposure on cognitive abilities. However, the administration of Selank demonstrated a remarkable reversal of these alcohol-induced cognitive deficits. Selank treatment significantly stimulated cognitive functions, leading to an increase in the exploration time of novel objects in both intact and alcohol-exposed rats. This suggests that Selank possesses genuine cognitive-enhancing properties and can effectively counteract the memory-impairing effects of chronic alcohol exposure [1].
Furthermore, the study shed light on the complex interplay between alcohol, BDNF, and Selank. The researchers observed that chronic alcohol consumption followed by withdrawal resulted in a significant increase in BDNF levels in both the hippocampus and prefrontal cortex. This initial surge in BDNF expression may represent an adaptive response to the damaging effects of ethanol, as the brain attempts to compensate for neuronal dysfunction and promote recovery. However, this compensatory increase in BDNF may not be sufficient to fully restore cognitive function.
The administration of Selank during alcohol withdrawal normalized BDNF content in both the hippocampus and prefrontal cortex, bringing it back to levels comparable to the control group. This suggests that Selank exerts its protective effects by modulating BDNF expression, preventing the excessive upregulation that occurs during alcohol withdrawal and restoring a more balanced state. The mechanism by which Selank regulates BDNF levels remains an area for further investigation. However, it is hypothesized that Selank may interact with the GABAergic system, which is known to be involved in the regulation of BDNF-related signaling pathways. By modulating GABAergic activity, Selank may indirectly influence BDNF expression and thereby protect against alcohol-induced cognitive deficits [1].
In conclusion, this research article provides compelling evidence that Selank, a peptide analogue of tuftsin, can effectively protect against ethanol-induced memory impairment by regulating BDNF content in the hippocampus and prefrontal cortex. These findings highlight the potential therapeutic value of Selank in treating cognitive deficits associated with chronic alcoholism [1].
2) The results of the study performed by Korakov demonstrate that Selank significantly impacts the spontaneous synaptic activity of hippocampal CA1 neurons in rats. Specifically, the application of Selank to hippocampal slices led to a marked increase in both the amplitude and discharge rate of spontaneous inhibitory postsynaptic currents (IPSCs) recorded in CA1 pyramidal neurons. This finding suggests that Selank enhances inhibitory neurotransmission within the hippocampus, a brain region critically involved in learning, memory, and anxiety regulation [2].
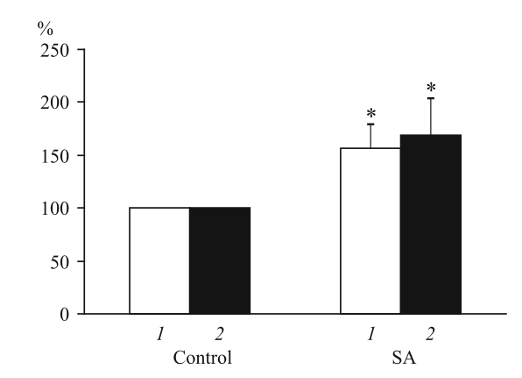
Figure 1: Effects of Selank versus a control compound on mean amplitude and discharge rates of IPSCs in hippocampal CA1 pyramidal neurons
The researchers observed this effect in a majority of the neurons tested, indicating a consistent and robust response to Selank. However, in a subset of neurons, the up-regulating effects of Selank on IPSCs were preceded by a short-term decrease in both the amplitude and discharge rate of these currents. This initial transient decrease lasted for approximately 1 to 1.5 minutes before the characteristic increase in IPSC activity was observed. The appearance of Selank’s effect was observed on minute six after its addition to the perfusion saline, indicating a delayed response [2].
To further characterize the effects of Selank, the researchers tested various concentrations of the drug ranging from 1 to 8 µM. Surprisingly, they found no significant dose-dependence within this concentration range. This implies that Selank exerts its effects on synaptic activity in a relatively saturable manner, with a maximal effect achieved even at the lower concentrations tested. This lack of dose-dependence may have implications for the therapeutic use of Selank, as it suggests that lower doses may be sufficient to achieve the desired effects.
Subsequent experiments were conducted to elucidate whether the observed effects of Selank resulted from up-regulation of secretion of the inhibitory neurotransmitter from the terminals of inhibitory interneurons or whether they reflected an increase in the somatic activity of these neurons. To address this question, the researchers blocked the transmission of nervous impulses along the axons of interneurons using tetrodotoxin (TTX). TTX pronouncedly decreased the amplitude and discharge rates of IPSCs, and Selank did not modulate these effects. This finding strongly suggests that the effects of Selank on spontaneous IPSCs in hippocampal CA1 pyramidal neurons are mediated by its action on interneurons, which directly innervate and regulate the activity of the pyramidal neurons. By modulating the activity of these interneurons, Selank indirectly influences the inhibitory tone within the hippocampus [2].
Furthermore, the biphasic character of Selank-induced changes in the amplitude and rate of spontaneous IPSCs was observed in some pyramidal neurons. These neurons experienced an initial low-grade drop in the number of high-amplitude IPSCs, and was followed by their pronounced elevation in comparison with control. This biphasic response can be explained by the asynchronous activation of several groups of inhibitory interneurons mutually intertwined with inhibitory neural projections.
In summary, the study’s results provide compelling evidence that Selank enhances inhibition of hippocampal CA1 pyramidal neurons by interneurons. These findings align with previous data on the mode of action of Noopept, a nootropic anxiolytic drug, suggesting a common mechanism for these types of drugs. The maintenance of a certain level of inhibition in the hippocampus, a structure playing a key role in the regulation of wakefulness, memory, and locomotion, seems to be a prerequisite for the realization of mnestic functions and optimal psychoemotional state. The study, supported by the Russian Science Foundation, contributes to a deeper understanding of the neuropharmacological mechanisms underlying the anxiolytic and nootropic effects of Selank [2].
Disclaimer
**LAB USE ONLY**
*This information is for educational purposes only and does not constitute medical advice. THE PRODUCTS DESCRIBED HEREIN ARE FOR RESEARCH USE ONLY. All clinical research must be conducted with oversight from the appropriate Institutional Review Board (IRB). All preclinical research must be conducted with oversight from the appropriate Institutional Animal Care and Use Committee (IACUC) following the guidelines of the Animal Welfare Act (AWA).
Citations
[1] Kolik LG, Nadorova AV, Antipova TA, Kruglov SV, Kudrin VS, Durnev AD. Selank, Peptide Analogue of Tuftsin, Protects Against Ethanol-Induced Memory Impairment by Regulating of BDNF Content in the Hippocampus and Prefrontal Cortex in Rats. Bull Exp Biol Med. 2019;167(5):641-644. doi:10.1007/s10517-019-04588-9
[2] Povarov IS, Kondratenko RV, Derevyagin VI, Myasoedov NF, Skrebitsky VG. Effect of Selank on Spontaneous Synaptic Activity of Rat Hippocampal CA1 Neurons. Bull Exp Biol Med. 2017;162(5):640-642. doi:10.1007/s10517-017-3676-3
[3] Falutz J, Mamputu JC, Potvin D, Moyle G, Soulban G, Loughrey H, Marsolais C, Turner R, Grinspoon S. Effects of tesamorelin (TH9507), a growth hormone-releasing factor analog, in human immunodeficiency virus-infected patients with excess abdominal fat: a pooled analysis of two multicenter, double-blind placebo-controlled phase 3 trials with safety extension data. J Clin Endocrinol Metab. 2010 Sep;95(9):4291-304. doi: 10.1210/jc.2010-0490. Epub 2010 Jun 16. PMID: 20554713.
Selank is a heptapeptide that is a synthetic derivative of the tetrapeptide tuftsin. Various animal studies show that Selank is able to act as an anxiolytic and an antidepressant. A study conducted by Sarkisova et. Al showed that Selank works by activating the monoaminergic systems in the brain, it helps to synthesize and release dopamine and regulate the activity of tyrosine hydroxylase.
The study used inbred WAG/Rij rats that had genetic-based symptoms of depression and compared them to BALB/c mice that had symptoms of situation-based depression in order to see whether the form of depression changed the effects that Selank had on the subjects. In order to determine whether or not Selank had an effect on depression in the test subjects, the animals underwent a forced swimming test as well as had their sucrose intake measured.
In high doses of 1000-2000 microg/kg, it was shown that Selank helped to reduce the symptoms of depression in the WAG/Rij rats. This was determined by their decreased sucrose intake and their increased immobilization during the forced swimming test. In low doses of 100 and 300 microg/kg, Selank was able to slightly reduce the time period of immobility in BALB/c mice but the effects were not as significant as the high doses of 600 and 900 microg/kg. Overall, the study concluded that there was enough evidence to suggest that Selank can help treat both genetic and situational causes of depression (https://pubmed.ncbi.nlm.nih.gov/18661785/).
Effects of Selank on Immune Responses
In addition to its positive effects on depression, Selank has been shown to help regulate immune responses and decrease inflammation. In a study conducted by Kolomin et. Al Selank and one of its fragments, Gly-Pro, was compared against the dynamics of various inflammatory genes such as C3, Casp1, Il2rg, and Xcr1 within the spleen of a mouse. The mice were given one intraperitoneal injection of Selank at a dose of 100 microg/kg. Following the injection, researchers found that Selank decreased C3 mRNA by three-fold only 30 minutes after the injection. Following the Selank injection, researchers reported a “wave-like” effect on the Casp1 mRNA, where levels would fluctuate back and forth. The research showed that there were significant changes made to the mRNA levels of both the Il2rg and the Xcr1 gene where expression was greatly reduced 90 minutes after the injection.
Overall this study concluded that treatment with Selank leads to the regulation of many of the genes that control various immune responses in the body, which indicates that Selank may play a role in modulating the immune system (https://www.sciencedirect.com/science/article/abs/pii/S0161589013005440).
PEPTIDES PREFER THE COLD
Keep peptide vials refrigerated at all times to reduce peptide bond breakdown. DO NOT FREEZE. Most peptides, especially shorter ones, can be preserved for weeks if careful.
Always swab the top of the vial with an alcohol wipe or rubbing alcohol before accessing.
ONLY MIX WITH STERILE BACTERIOSTATIC WATER
The purity and sterility of bacteriostatic water are essential to prevent contamination and to preserve the shelf-life of dissolved peptides.
Push the pin through the rubber stopper at a slight angle, so that you inject the bacteriostatic water toward the inside wall of the vial, not directly onto the powder.
Lyophilized peptide should be stored at -20°C (freezer), and the reconstituted peptide solution at 4°C (refrigerated). Do not freeze once reconstituted.
NEVER SHAKE A VIAL TO MIX.
Air bubbles are unfavorable to the stability of proteins.
Selank is sold for laboratory research use only. Terms of sale apply. Not for human consumption, nor medical, veterinary, or household uses. Please familiarize yourself with our Terms & Conditions prior to ordering.




Valoraciones
No hay valoraciones aún.