Descripción
PT-141 (Bremelanotide) Peptide Vial
| CAS Number | 189691-06-3 |
| Other Names | Bremelanotide, UNII-6Y24O4F92S, PT141, 6Y24O4F92S, Vyleesi (TN) |
| IUPAC Name | (3S,6S,9R,12S,15S,23S)-15-[[(2S)-2-acetamidohexanoyl]amino]-9-benzyl-6-[3-(diaminomethylideneamino)propyl]-12-(1H-imidazol-5-ylmethyl)-3-(1H-indol-3-ylmethyl)-2,5,8,11,14,17-hexaoxo-1,4,7,10,13,18-hexazacyclotricosane-23-carboxylic acid |
| Molecular Formula | C50H69N15O10 |
| Molecular Weight | 1040.2 |
| Purity | ≥99% Pure (LC-MS) |
| Liquid Availability | N/A |
| Powder Availability | N/A |
| Storage Condition | Store cold, keep refrigerated. Do NOT freeze. |
| Terms | All products are for laboratory developmental research USE ONLY. Products are not for human consumption. |
**Información Importante: Cada péptido se entrega liofilizado y debe ser reconstituido con Agua Bacteriostática para poder ser administrado en forma líquida.
Vea aquí el video sobre la reconstitución de péptidos
What is Bremelanotide?
Bremelanotide is a synthetic peptide originally developed for its potential to treat sexual dysfunction. It is a melanocortin receptor agonist that activates specific receptors in the brain, leading to increased arousal. Initially investigated for its applications in restoring sexual function, bremelanotide has gained recognition for its unique mechanism of action, distinguishing it from other therapies. Development of this compound represents a significant advancement in peptide-based pharmacology aimed at addressing sexual health issues.
Ver más…
3 Main Research Findings
1) Bremelanotide was found to be well tolerated and safe, demonstrating the potential to significantly improve sexual desire and related distress in premenopausal women.
2) Varying doses of bremelanotide was shown to be safe and effective at treating female sexual dysfunction in premenopausal women.
Selected Data
1) The studies conducted by the research team of Kingsberg et al consisted of two identical, randomized, double-blind, placebo-controlled trials conducted across multiple centers to evaluate the safety and efficacy of bremelanotide administered via subcutaneous injection as needed in premenopausal women with hypoactive sexual desire disorder (HSDD). The trial design included a core study phase consisting of a four-week screening period, followed by a four-week baseline phase where participants received only a placebo in a single-blind manner, and then a 24-week randomized treatment phase, where participants received either bremelanotide or placebo [1].
Additionally, an optional 52-week open-label extension was available for women who completed the core study. The target sample size was approximately 1,500 women per study, with the goal of enrolling around 550 women for randomization, and collecting over two months of data from at least 450 women during the double-blind treatment phase. Sample size calculations were based on phase 2 trial data, which predicted a treatment difference of 1.0 in the Female Sexual Function Index-desire domain (FSFI-D) score and about 20.43 points in the Female Sexual Distress Scale-Desire/Arousal/Orgasm (FSDS-DAO) item 13, with a total of 450 evaluable patients providing sufficient statistical power to detect significant differences.
Participants were randomized in a 1:1 ratio to receive either 1.75 mg of bremelanotide or a placebo, using an interactive response system and permuted blocks stratified by country. The initial phase involved a four-week screening followed by a four-week single-blind placebo run-in period, during which all women were required to engage in at least one sexual activity to establish baseline data and reduce placebo effects. Those who completed the placebo run-in were then randomized to respective treatment arms for the 24-week double-blind phase. The 1.75 mg bremelanotide dose was chosen based on earlier dose-finding studies, demonstrating optimal efficacy and safety. During the treatment phase, women could self-administer up to 12 doses in each four-week interval, about 45 minutes prior to anticipated sexual activity, with no more than one dose per 24 hours [1].
Throughout the study, safety assessments included physical examinations, vital sign measurements, electrocardiograms, laboratory testing, adverse event monitoring, and the administration of the Beck Scale for Suicidal Ideation. These evaluations occurred at screening, monthly during the core phase, and at the time of study completion or early discontinuation. Diagnosis of HSDD was confirmed using a standardized screening guide, and participants were required to have experienced acquired, generalized HSDD for more than six months, with prior normal sexual function for at least two years, and an ongoing willingness to engage in sexual activity at least once a month. Women who were pregnant, nursing, had other female sexual dysfunctions, or had recent psychiatric diagnoses or substance use issues were excluded from the studies. Patients using medications such as neuroleptics, antidepressants, mood stabilizers, and others within specified time frames before screening were also disqualified [1].
The primary endpoints focused on measuring changes from baseline to the end of the study in two key areas: the FSFI-D score, which assesses sexual desire based on specific questions, and the FSDS-DAO item 13, which gauges feelings of being bothered by low desire. A secondary endpoint involved the number of satisfying sexual events occurring within 16 hours of taking the study drug and reported within 72 hours. The primary efficacy analysis employed a last-observation-carried-forward approach within a modified intent-to-treat population, which included all participants who had at least one follow-up visit after randomization. A safety population comprised all women who received at least one dose of the assigned treatment.
An independent assessment committee evaluated the endpoints and determined threshold criteria for clinically meaningful responses before the trial’s unblinding. Since both studies were conducted under similar protocols, data from both were combined and reported jointly. Overall, the studies aimed to rigorously assess whether bremelanotide could provide a meaningful improvement in sexual desire and distress among women with HSDD, with safety and tolerability also closely monitored throughout the trial periods [1].
2) This study performed by the research team of Clayton et al focused on female sexual dysfunction (FSD), particularly among premenopausal women diagnosed with conditions such as Female Sexual Arousal Disorder (FSAD), Hypoactive Sexual Desire Disorder (HSDD), or a combination of both. The study aimed to evaluate the efficacy and safety of a subcutaneous drug called bremelanotide (BMT) at different dosage levels compared to placebo. Participants included women at least 21 years old who were nonpregnant, had persistent sexual desire or arousal issues for at least six months, and demonstrated a prior history of sexual functionality spanning at least two years. To qualify, these women also had to be involved in a current stable, monogamous relationship lasting a minimum of six months, with a commitment to be sexually active at least once monthly. If a participant changed partners during the trial, she was discontinued from the study [2].
Exclusion criteria were extensive, designed to eliminate women with unstable or uncontrolled health conditions or those whose sexual problems could be attributed to other factors. Women with untreated or recent pelvic inflammatory disease, cervical dysplasia, significant cervicitis, urinary tract infections, or chronic dyspareunia within the previous year were excluded. Additionally, women with active moderate to severe vaginitis or other significant infections, gynecologic conditions affecting study procedures, unresolved sexual trauma or abuse histories, or prior hysterectomies involving menstrual cycle alterations were not eligible. The study also excluded women with sexual dysfunction secondary to untreated endocrine diseases, those currently undergoing psychotherapy for sexual dysfunction, or those who had recent treatment for depression or psychosis. The use of antidepressants or antipsychotics within the last three months was also grounds for exclusion. Other exclusion criteria included the use of any medication or nutritional supplement affecting sexual desire or arousal, uncontrolled hypertension (defined by specific blood pressure thresholds), recent changes in antihypertensive therapy, or other serious medical conditions [2].
The study followed a structured design; a one-month no-treatment phase was used to confirm diagnoses. Afterward, each woman self-administered a single-blind, placebo dose in a clinical setting, supervised by research staff, followed by a one-week monitoring period. This was succeeded by a four-week at-home placebo phase to establish each participant’s baseline sexual activity. Randomization occurred after baseline assessments, with participants assigned in a 1:1:1:1 ratio to receive either a placebo or one of three BMT doses including 0.75, 1.25, or 1.75 mg. Randomization was stratified based on diagnosis of either FSAD, HSDD, or mixed. The intervention began with two weekly in-clinic doses, after which participants self-administered the drug at home approximately one week apart, around 45 minutes prior to anticipated sexual activity, with a maximum of one dose daily and up to 16 doses over four weeks.
Throughout the intervention phase, participants were assessed every four weeks to monitor treatment effects and safety. The study drug was supplied in prefilled syringes for subcutaneous injection into the thigh or abdomen. The primary outcome measure was the change in the number of satisfactory sexual encounters, as reported through question 10 (Q10) of an adapted questionnaire called the Female Sexual Encounter Profile-Revised (FSEP-R). Participants recorded whether each sexual encounter was satisfactory, and the primary end point was calculated as the difference between the number of satisfactory encounters during the last four weeks of treatment and those recorded at baseline. To confirm efficacy, the measure focused only on the last four weeks of the intervention period, with data collected electronically within 24 hours after each sexual event [2].
Secondary outcomes involved multiple questionnaires assessing various aspects of sexual function. These included the Female Sexual Function Index (FSFI), a comprehensive 19-item self-assessment measuring desire, arousal, lubrication, orgasm, satisfaction, and pain, with higher scores indicating better function. Changes from baseline to study end in total FSFI scores and specific domain scores of desire and arousal were analyzed. Also evaluated were the Female Sexual Distress Scale-Desire/Arousal/Orgasm (FSDS-DAO), a 15-item measure where lower scores indicated improvement; and individual items related to desire and arousal. These assessments helped provide a thorough understanding of how the treatment affected different facets of sexual health.
Safety assessments were a key component, with ongoing monitoring for adverse events throughout the trial. Participants reported any negative effects, and safety evaluations included physical exams, vital signs, electrocardiograms, and blood tests. Blood pressure monitoring was particularly intensive because of prior concerns about potential cardiovascular effects associated with similar formulations. Blood pressure was manually checked during the initial in-clinic doses and continuously monitored via 24-hour ambulatory BP measurement (ABPM), with readings taken every 15 minutes. Patients were withdrawn if they experienced sustained elevations in blood pressure beyond predefined thresholds or significant increases from baseline [2].
Discussion
1) The results collected from the two trials conducted by the research team of Kingberg et al demonstrated that women treated with bremelanotide showed statistically significant improvements in sexual desire and reductions in distress related to low desire compared to placebo. Specifically, FSFI-D scores increased by 0.30 in study 301 and 0.42 in study 302, and the FSDS-DAO scores decreased by 0.37 and 0.29, respectively. When data from both studies were combined, the overall effect size for improving desire was 0.39, while the reduction in distress was 0.27, both favoring bremelanotide. These benefits were evident beginning at week 4 and persisted throughout the duration of the studies. The cumulative distribution analysis of the primary endpoints revealed highly significant differences between treatment groups, favoring bremelanotide [1].
Support for clinical significance came from a responder analysis based on an anchored assessment, which evaluated participants’ perception of benefit. Approximately 58% of women in the bremelanotide group reported meaningful benefits, compared to about 36% in the placebo group across both studies. Additional sensitivity analyses, including three static anchor assessments and assumptions that early discontinuers were nonresponders, confirmed the robustness of these findings.
Overall sexual functioning and distress levels were further examined through total scores on the FSFI and FSDS-DAO scales. The odds ratios for responder status was defined as a score of 4.2 or higher for FSFI or -10 or lower for FSDS-DAO, and was shown to favor bremelanotide. This indicates a higher likelihood of meaningful improvement in desire and associated distress. The trends observed across both studies consistently showed more responders in the bremelanotide group for secondary measures involving desire, arousal, and satisfaction [1].
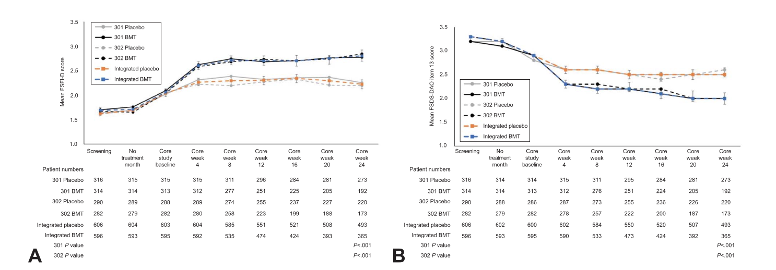
Figure 1: Changes in A) FSFI-D scores, and B) FSDS-DAO scores following treatment with bremelanotide.
2) This study performed by researchers Clayton et al offers a comprehensive overview of the demographic and baseline characteristics of participants, as well as the efficacy and safety outcomes observed during the clinical study investigating the effects of BMT, a potential treatment for female sexual dysfunction (FSD). The participant characteristics are first summarized, followed by detailed efficacy results, exploratory analyses, and safety monitoring data [2].
As for the baseline characteristics of all women receiving the double-blind study drug, overall, the average age of participants was 37 years, with an age range from 21 to 53 years. Most of the women were white, accounting for 71% of the population. The majority experienced regular menstrual cycles, with 79% reporting regular menses. Additionally, 87% of participants were not using oral contraceptives at baseline. Regarding their sexual health diagnoses, 74% of the modified intent-to-treat (mITT) population had both FSAD and HSDD, while 23% had only HSDD and 3% had only FSAD. Baseline measures of sexual function show that during the four weeks immediately preceding randomization, women reported an average of 1.5 to 1.9 satisfactory sexual events (SSEs) per month across treatment groups. These initial data establish a relatively homogenous study population with similar baseline levels of sexual activity [2].
Turning to efficacy outcomes, the results demonstrate that BMT treatment produced statistically significant and clinically meaningful improvements in both the primary and many secondary endpoints. In the mITT population, the average change in the number of SSEs from baseline to the end of the study was +0.7 events per month in the pooled groups receiving 1.25 and 1.75 mg doses of BMT, compared to only +0.2 events in the placebo group. The difference between the pooled BMT doses and placebo was statistically significant, indicating that women treated with higher doses of BMT experienced a notable increase in sexual satisfaction compared to those on placebo.
Secondary efficacy measures further support the treatment’s benefits. The FSFI, a validated 19-item questionnaire assessing various domains of female sexual function, showed a mean increase of 3.6 points in total score for the BMT group, compared to a 1.9-point increase in the placebo group. This indicates that BMT contributed to significant improvements in overall sexual function. Similarly, the FSDS-DAO, which measures sexual distress with lower scores reflecting less distress, demonstrated a greater reduction in scores among BMT-treated women, with an average decrease of 11.1 points, versus a 6.8-point decrease in placebo recipients. This shows that BMT not only increased sexual activity but also reduced associated distress [2].
Analyzing the responder proportion, that is, the percentage of women showing a clinically significant response, provided further evidence of BMT’s effectiveness. Using historical benchmarks for meaningful change, significantly higher proportions of women in the pooled 1.25/1.75 mg BMT groups responded on key endpoints. Specifically, 51% of women in the pooled BMT dose groups experienced an increase in SSEs per month compared to 37% in the placebo group. For improvements in FSFI total scores, 47% of women in the BMT group responded versus only 29% on placebo. The proportion of women who reported decreased sexual distress as measured by FSDS-DAO was 64% in the BMT group versus 45% in placebo. These responder analyses strongly support the clinical relevance of the observed improvements [2].
Data visualization emphasized that women treated with the higher BMT doses experienced consistent improvements over baseline. However, it was highlighted that some measures did not reach statistical significance but overall trends favored BMT. For example, the arousal score of the FSDS-DAO did not show a significant change, which rendered all secondary endpoints exploratory and p-values nominal. Nonetheless, significant improvements compared to placebo were observed in the total FSDS-DAO and FSFI scores, particularly at the 1.75 mg dose, which was associated with improvements across multiple sexual health domains.
An exploratory assessment of episodic responses further explored the effects of BMT at the individual encounter level. Participants provided post-encounter responses on their arousal, desire, and satisfaction levels through the Female Sexual Encounter Profile-Revised (FSEP-R). Averaged over a 4-week period at the end of the study, women receiving 1.25 mg and 1.75 mg of BMT demonstrated statistically significant improvements in their ratings of sexual arousal, satisfaction with arousal, sexual desire, and satisfaction with desire. These findings suggest that BMT not only improves overall sexual function but also enhances the quality of individual sexual experiences [2].
The study also conducted exploratory analyses focused on women with HSDD as a primary or sole diagnosis, which represented approximately 86% of the study population. The baseline characteristics and sexual function measures of these women closely resembled those of the entire group. At the end of the study, women with HSDD experienced similar improvements in the number of SSEs per month and in FSFI and FSDS-DAO scores, aligning with the overall study outcomes. These results support the potential efficacy of BMT specifically for women diagnosed primarily with HSDD.
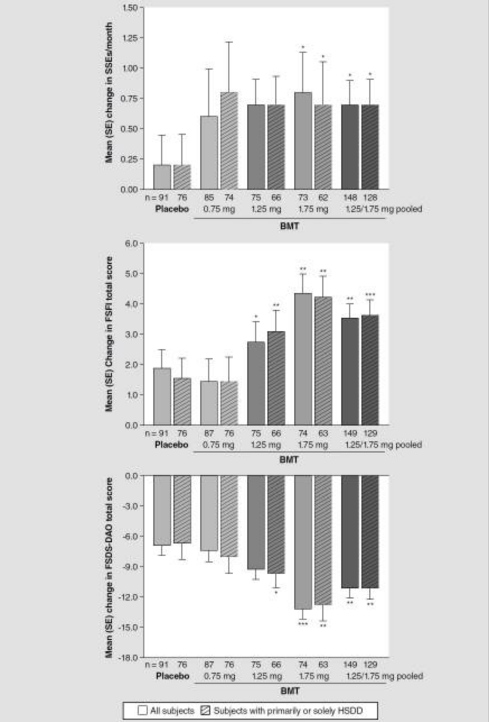
Figure 2: Changes in mean number of SSEs per month, mean changes in FSFI total score, and mean changes in FSDS-DAO scores.
In conclusion, the findings from this clinical trial demonstrate that subcutaneous BMT at doses of 1.25 mg and 1.75 mg can significantly improve sexual function, reduce sexual distress, and enhance sexual experience for women suffering from FSD, with a generally favorable safety profile. The improvements were clinically meaningful, statistically significant, and observed across multiple measures of sexual health, including frequency of satisfactory sexual events, overall sexual functioning scores, and distress levels. Safety monitoring confirmed transient blood pressure changes that did not lead to any serious adverse events, supporting the potential viability of BMT as a treatment option for women with sexual desire and arousal concerns [2].
Disclaimer
**LAB USE ONLY**
*This information is for educational purposes only and does not constitute medical advice. THE PRODUCTS DESCRIBED HEREIN ARE FOR RESEARCH USE ONLY. All clinical research must be conducted with oversight from the appropriate Institutional Review Board (IRB). All preclinical research must be conducted with oversight from the appropriate Institutional Animal Care and Use Committee (IACUC) following the guidelines of the Animal Welfare Act (AWA).
Citations
[1] Kingsberg SA, Clayton AH, Portman D, et al. Bremelanotide for the Treatment of Hypoactive Sexual Desire Disorder: Two Randomized Phase 3 Trials. Obstet Gynecol. 2019;134(5):899-908. doi:10.1097/AOG.0000000000003500
[2] Clayton AH, Althof SE, Kingsberg S, et al. Bremelanotide for female sexual dysfunctions in premenopausal women: a randomized, placebo-controlled dose-finding trial. Womens Health (Lond). 2016;12(3):325-337. doi:10.2217/whe-2016-0018
PT-141, also known as Bremelanotide, is a peptide that has been shown to have positive effects on rats and primates in helping erectile dysfunction and various other forms of sexual dysfunction. Bremelanotide is shown to be an agonist at melanocortin receptors such as MC3R and MC4R and is also considered to be a melanocyte-stimulating hormone that is an active metabolite of Melanotan II. Various studies have begun to show how melanocortins have the ability to positively or negatively affect sexual health.
Bremelanotide is derived from the peptide hormone, melanotan II. It was originally recognized that melanocyte receptor stimulators play a role in sexual behavior in the 60s when research showed that the introduction of these receptors caused arousal in male rats. From there, researchers developed analogs such as melanton I and II. Melanotan II was further specialized and reduced into the derivative Bremelanotide. Bremelanotide is meant to bind to MC3R and MC4R has been shown to increase levels of arousal in both male and female rodents through the nervous system rather than the circulatory system (https://vitality-sciences.com/peptides/pt-141-erectile-dysfunction/).
In a study conducted by Semple et. Al, rodents with a knockout melanocortin 4 receptor (MC4R) were observed to determine how levels of sexual dysfunction varied without a proper functioning melanocortin pathway. The study found that the rodents that had this knockout gene displayed increased levels of obesity, decreased libido, and an inability to copulate. In order to explain why these phenomena were occurring, researchers reintroduced MC4R into the neurocircuitry on Sim 1 neurons and oxytocin neurons.
By reintroducing MC4R to the Sim 1 neurons it was found that latency to ejaculation as well as overall changes in sexual behavior were far lower than the mice with the knockout gene. This indicates that when MC4R is expressed on Sim 1 neurons, negative changes in sexual behavior are combatted. On a similar note, when MC4R was reintroduced to the oxytocin neurons there was a similar decrease in ejaculation latency and changes in sexual behavior (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6684847/).
Bremelanotide Increases Female Sex Drive in Rats
A study conducted by Molinoff et. Al, Bremelanotide was administered to primates and rats and showed a significant decrease in erectile dysfunction in the male of the species. It was shown through c-Fos immunoreactivity that systemic introduction of Bremelanotide has been shown to activate neurons found in the hypothalamus. Due to the involvement of melanocyte receptors within the hypothalamus, it is indicated that the hypothalamus is crucial to proper sexual functioning and behaviors (https://pubmed.ncbi.nlm.nih.gov/12851303/).
A study conducted by researchers from Palatin Technologies Inc. examined the effects Bremelanotide had on female rats. Prior to sexual activity, female rats exhibit certain behaviors in order to arouse the male of the species. The behaviors of each experimental group were compared to the control group of female rats. It was discovered that after administering various doses of Bremelanotide to female rats there was an overall increase in the number of different sexual behaviors they were exhibiting before copulation in a dose-dependent manner. Furthermore, the females that had received treatment with Bremelanotide were also more willing to seek out sexual activity and had more of a desire to copulate with the male rats (https://www.eurekalert.org/news-releases/837832).
PEPTIDES PREFER THE COLD
Keep peptide vials refrigerated at all times to reduce peptide bond breakdown. DO NOT FREEZE. Most peptides, especially shorter ones, can be preserved for weeks if careful.
Always swab the top of the vial with an alcohol wipe or rubbing alcohol before accessing.
ONLY MIX WITH STERILE BACTERIOSTATIC WATER
The purity and sterility of bacteriostatic water are essential to prevent contamination and to preserve the shelf-life of dissolved peptides.
Push the pin through the rubber stopper at a slight angle, so that you inject the bacteriostatic water toward the inside wall of the vial, not directly onto the powder.
Lyophilized peptide should be stored at -20°C (freezer), and the reconstituted peptide solution at 4°C (refrigerated). Do not freeze once reconstituted.
NEVER SHAKE A VIAL TO MIX.
Air bubbles are unfavorable to the stability of proteins.
PT-141 is sold for laboratory research use only. Terms of sale apply. Not for human consumption, nor medical, veterinary, or household uses. Please familiarize yourself with our Terms & Conditions prior to ordering.




Valoraciones
No hay valoraciones aún.