Descripción
AOD9604 Peptides Vials
| CAS Number | 221231-10-3 |
| Other Names | AOD9604 acetate, AOD 9604 acetate, AOD-9604 acetate |
| IUPAC Name | acetic acid;(2S)-2-[[2-[[(4R,7S,13S,16S,19S,22S,25R)-25-[[(2S)-5-amino-2-[[(2S)-2-[[(2S,3S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]-4-methylpentanoyl]amino]-5-carbamimidamidopentanoyl]amino]-3-methylpentanoyl]amino]-3-methylbutanoyl]amino]-5-oxopentanoyl]amino]-22-(3-carbamimidamidopropyl)-13-(2-carboxyethyl)-7,19-bis(hydroxymethyl)-6,9,12,15,18,21,24-heptaoxo-16-propan-2-yl-1,2-dithia-5,8,11,14,17,20,23-heptazacyclohexacosane-4-carbonyl]amino]acetyl]amino]-3-phenylpropanoic acid |
| Molecular Formula | C80H127N23O25S2 |
| Molecular Weight | 1875.1 |
| Purity | ≥99% Pure (LC-MS) |
| Liquid Availability | N/A |
| Powder Availability | N/A |
| Storage Condition | Store cold, keep refrigerated. Do NOT freeze. |
| Terms | All products are for laboratory developmental research USE ONLY. Products are not for human consumption. |
**Información Importante: Cada péptido se entrega liofilizado y debe ser reconstituido con Agua Bacteriostática para poder ser administrado en forma líquida.
Vea aquí el video sobre la reconstitución de péptidos
What is AOD9604?
AOD9604 is a synthetic peptide fragment derived from human growth hormone, specifically encompassing amino acids 176–191 of the hormone’s sequence. Designed to retain the fat-reducing effects of HGH without influencing growth or insulin-like growth factor 1 levels, AOD9604 has gained attention for its potential as an anti-obesity and metabolic treatment. By mimicking the lipolytic activity of HGH while avoiding the associated anabolic or mitogenic effects, AOD9604 offers a targeted approach to adipose tissue regulation. Current research findings suggest that it may promote fat loss, enhance lipid metabolism, and improve overall metabolic health with a favorable safety profile.
Ver más…
3 Main Research Findings
1) Intra-articular administration of AOD9604 was shown to improve cartilage regeneration in cases of collagenase-induced knee osteoarthritis.
2) Treatment with AOD9604 has been found to increase expression of beta-3-AR RNA, indicating that the compound has the potential to enhance lipolytic sensitivity.
Selected Data
1) The research study conducted by the research team of Kwon and Park aimed to evaluate the protective effects of the peptide AOD9604 when administered via intra-articular injection on cartilage in a collagenase-induced osteoarthritis (OA) rabbit model. Additionally, the study assessed whether combining AOD9604 with hyaluronic acid (HA) would enhance therapeutic outcomes. To accomplish this, researchers used 32 male New Zealand white rabbits, each 12 weeks old. These animals were housed under controlled environmental conditions and had free access to food and water [1].
To induce OA, the rabbits were first anesthetized using a combination of xylazine and ketamine. Once anesthetized, the right knee joints of the animals were shaved, sterilized, and subjected to an intra-articular injection of collagenase type II. The enzyme was dissolved in phosphate-buffered saline at pH 7.4, sterilized through a 0.22-μm membrane, and administered under ultrasound guidance. A second collagenase injection was administered three days later. This two-step injection protocol is known to induce OA-like cartilage degeneration, particularly in the lateral portion of the femoral condyle.
Following OA induction, the rabbits were randomly divided into four groups with 8 subjects, each receiving a different weekly intra-articular treatment starting four weeks after the first collagenase injection. Group 1 received a saline injection as a control. Group 2 was treated with HA, at a molecular weight of 3.0×10⁶ Da and a concentration of 10 mg/mL, delivering a dose of 6 mg per injection. Group 3 received 0.25 mg of AOD9604 per 0.6 mL injection, and Group 4 received a combination of AOD9604 at 0.25 mg and HA at 6 mg. All injections were performed under general anesthesia using ultrasound guidance by a trained physiatrist under sterile conditions. After the injection regimen, no further medication was given. The rabbits were euthanized via carbon monoxide inhalation nine weeks after the first collagenase injection for subsequent examination [1].
Clinical evaluation included daily assessment of lameness, defined as the inability of the rabbit to bear weight and maintain the typical flexion-extension cycle of the affected limb compared to the unaffected limb. Each rabbit was observed individually in a controlled ground area for 20 minutes daily at a set time. The time required for each rabbit to regain normal ambulation, without signs of lameness, was recorded. These observations were performed by three independent physiatrists who were blinded to the treatment groups, ensuring unbiased assessment.
After euthanasia, the knee joints were surgically exposed, and the femoral and tibial condyles were examined for gross morphological changes. The researchers focused on the lateral femoral condyle, where the collagenase injection had caused the most significant damage. Cartilage degeneration was scored using the Yoshimi scale, which ranges from 0, defined as normal cartilage, to 5, defined as complete loss of cartilage. This scoring system allowed for semi-quantitative evaluation of cartilage integrity based on macroscopic features such as softening, fibrillation, erosion, ulceration, and complete cartilage loss [1].
For histological analysis, the femoral and tibial condyles were fixed in 10% neutral buffered formalin, decalcified with 20% EDTA, embedded in paraffin, and sectioned into 5-μm thick frontal slices. These slices were stained with hematoxylin and eosin, focusing on the lateral portion of the femoral condyle. If initial staining was insufficient, the sample was sectioned again to obtain a better visual field of cartilage structure. Histopathological evaluation used a modified Mankin scoring system. This system rated cartilage structural integrity from 0, defined as normal to 6, defined as complete disorganization, and assessed cellularity from 0, defined as normal, to 3, defined as hypocellularity, for a total possible score of 0 to 9. Higher scores indicated more severe degeneration [1].
Statistical analysis was conducted using SPSS version 14.0. The Kruskal–Wallis test was used to evaluate differences in gross morphological scores, histopathological scores, and lameness duration across all four treatment groups. For comparisons between two specific groups, the Mann–Whitney U test was applied. A p-value less than 0.05 was considered statistically significant, indicating a meaningful difference in treatment efficacy. This study was designed to systematically investigate the cartilage-protective effects of AOD9604, both alone and in combination with HA, using a well-established collagenase-induced OA rabbit model. The experiment included detailed clinical, morphological, and histological assessments to determine the degree of joint degeneration and recovery following different intra-articular treatments [1].
2) This study conducted by the research team of Heffernan et al investigated the biological effects of AOD9604, a synthetic peptide fragment of HGH, in various mouse models, including lean and obese mice, as well as wild-type (WT) and β3-adrenergic receptor knockout (β3-KO) mice. AOD9604 was specifically designed to include amino acid residues 177–191 of hGH with an additional tyrosine residue at the N-terminus. The peptide was synthesized using solid-phase methods and purified via reverse-phase high-performance liquid chromatography (HPLC). Its structure was confirmed by mass spectrometry and amino acid analysis [2].
The experiment involved several treatment groups across multiple animal models. Lean C57BL/6J mice and genetically obese (ob/ob) mice, all 12 weeks old, were divided into three treatment groups including: saline control, AOD9604 at a dose of 250 µg/kg/day, and hGH at a dose of 1 mg/kg/day, with each group consisting of six mice. The animals were kept in a controlled environment with stable temperature and humidity under a 12-hour light/dark cycle. The compounds were intraperitoneally administered with the compound at 8 a.m. daily for 14 days using a 1-mL syringe and a 23-gauge needle. Body weight and food intake were recorded every other day.
After 14 days of treatment, the mice were euthanized via cardiac injection of sodium pentobarbitone given at 35 mg/kg. Their white epididymal and brown interscapular adipose tissues were dissected, weighed, and frozen in liquid nitrogen for later RNA analysis. In addition to the lean and ob/ob mice, the study included β3-KO mice and WT controls obtained from genetically verified breeding colonies. These animals were also subjected to the same environmental conditions and treatment protocols for a chronic 28-day administration study [2].
In the β3-KO and WT groups, male mice aged 12–14 weeks were divided into three subgroups including: saline control, AOD9604 administered at a dose of 250 µg/kg/day, and hGH administered at a dose of 1 mg/kg/day. Each subgroup included three to five animals. On day 0, blood samples were collected via anesthesia-induced cardiac puncture and stored for plasma glycerol analysis. Daily intraperitoneal injections of AOD9604 or hGH were then administered for 28 days, during which time body weight and food intake were recorded every other day. At the end of the study, final blood samples were taken, and the animals were euthanized by cardiac injection. Their adipose tissues were collected and weighed for further analysis [2].
Plasma glycerol levels were determined enzymatically using a glycerol phosphate oxidase assay kit and measured with a spectrophotometer. The results were expressed in micromoles per deciliter and reported as changes from the baseline values. RNA extraction and gene expression analysis were also integral to this study. Tissue samples from lean and ob/ob mice were homogenized in TRIzol reagent, followed by RNA extraction using standard protocols. The isolated RNA was quantified spectrophotometrically using the A260/A280 ratio. Complementary DNA was synthesized from 1 µg of total RNA through reverse transcription; this process involved denaturation, enzyme reaction, and thermal cycling to produce stable complementary DNA for subsequent analysis.
PCR amplification targeted the β3-adrenergic receptor (β3-AR) gene and β-actin as a housekeeping control. Specific oligonucleotide primers for β3-AR and actin were used, generating PCR products of 234 base pairs and 559 base pairs, respectively. To detect any contamination by genomic DNA, the primers were designed to span intron-exon boundaries. Radiolabeled reverse primers were prepared and incorporated into PCR reactions to facilitate detection and quantification. After amplification, PCR products were electrophoresed on agarose gels and transferred onto nylon membranes using Southern blotting techniques. The membranes were hybridized and exposed to phosphor screens for imaging. Band intensities corresponding to β3-AR were normalized to β-actin expression using MCID imaging software. The expression levels were compared between treated and control groups [2].
Overall, the study demonstrated that AOD9604 affects metabolic parameters in both lean and obese mice, as well as in genetically modified mouse models lacking β3-adrenergic receptors. Compared to hGH, AOD9604 offered similar adipose tissue effects without significantly increasing systemic growth factors, such as IGF-1. The study also highlighted AOD9604’s influence on fat metabolism and energy expenditure, and its interaction with β3-adrenergic pathways [2].
Discussion
1) The experiment performed by researchers Kwon and Park investigated the therapeutic potential of AOD9604, a synthetic peptide fragment of HGH, in a rabbit model of OA. Specifically, researchers examined the effects of intra-articular injections of AOD9604, both alone and in combination with HA, on cartilage morphology, histopathology, and functional outcomes such as lameness. The findings showed that rabbits treated with AOD9604 demonstrated improved joint outcomes compared to control animals, with less morphological and histopathological cartilage damage. Notably, the combination of AOD9604 and HA appeared to offer greater benefits than either agent alone, suggesting a synergistic effect in promoting cartilage protection and repair [1].
AOD9604 is a disulfide-constrained peptide composed of 15 amino acids derived from the C-terminal end of human GH, with an added tyrosine residue at the N-terminal. Despite being a GH fragment, AOD9604 does not bind with high affinity to the HGH receptor and does not promote cell proliferation in HGH receptor-transfected cells. Nonetheless, it retains some biological activities associated with HGH, such as stimulating cartilage metabolism and potentially influencing chondrocyte function. These properties are relevant given the established role of HGH in cartilage growth and repair through the local stimulation of IGF-1.
Initially developed for obesity treatment, AOD9604 showed weight-reducing effects in rodent models comparable to those of full-length GH, but without inducing diabetes or stimulating IGF-1 production. In vitro studies revealed that AOD9604 enhances the differentiation of adipose-derived mesenchymal stem cells into bone, stimulates the production of proteoglycans and collagen in chondrocytes, and promotes myoblast differentiation into muscle cells. These actions align with the types of tissue repair in bone, cartilage, and muscle that are essential in managing OA [1].
Although previous studies had examined the effects of intra-articular GH injections on cartilage repair, few had compared the use of GH analogs or peptides like AOD9604 in combination with HA. In this study, both AOD9604 and HA individually led to improved cartilage outcomes and shorter periods of lameness compared to saline control. However, the group receiving both AOD9604 and HA showed the most favorable results, suggesting a potential synergistic interaction. HA may exert a chondroprotective effect through anti-inflammatory properties and improved joint lubrication, while AOD9604 may activate developmental pathways that support cartilage regeneration. These findings align with earlier research showing that combining HA with GH enhances cartilage repair more effectively than HA alone [1].
The dosage of AOD9604 used in this study or 0.25 mg per injection, was carefully calculated to be molar equivalent to doses of full-length GH shown to be effective in prior rabbit OA studies. Specifically, 3 mg of GH in 0.6 mL is approximately equivalent, on a molar basis, to 0.25 mg of AOD9604. Additionally, the concentration of AOD9604 in the synovial fluid after injection was estimated to be about 0.19 mg/mL, similar to concentrations used in previous GH injection studies in animal models.
One important factor influencing the efficacy of intra-articular drugs is their residence time within the joint. AOD9604, when injected in saline, may be rapidly cleared from the joint due to uptake into local circulation, limiting its therapeutic effects. However, when combined with HA, the drug may remain longer in the joint cavity, increasing bioavailability and therapeutic potential. This is consistent with studies showing that HA can improve the performance of nanoparticle-based drug formulations by extending local retention and reducing systemic absorption [1].
HA also contributes directly to cartilage protection through its high molecular weight, which guards chondrocytes against oxidative stress caused by reactive oxygen species. In this study, the high molecular weight HA used in combination with AOD9604 may have further supported chondrocyte survival and matrix synthesis, contributing to the observed improvements in cartilage morphology and function.
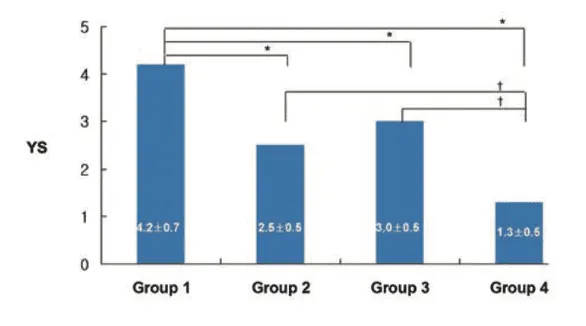
Figure 1: Changes in gross morphological scores amongst the four experimental treatment groups.
The biological plausibility of AOD9604’s cartilage-regenerating effects is further supported by insights from conditions such as acromegaly, where excess GH leads to abnormally thick cartilage due to elevated local IGF-1 production. However, chronic GH treatment carries substantial risks, including glucose intolerance, insulin resistance, diabetes, cancer, and cardiovascular complications. Because AOD9604 does not stimulate IGF-1 production and lacks strong GH receptor agonism, it may present a safer alternative for long-term OA treatment [1].
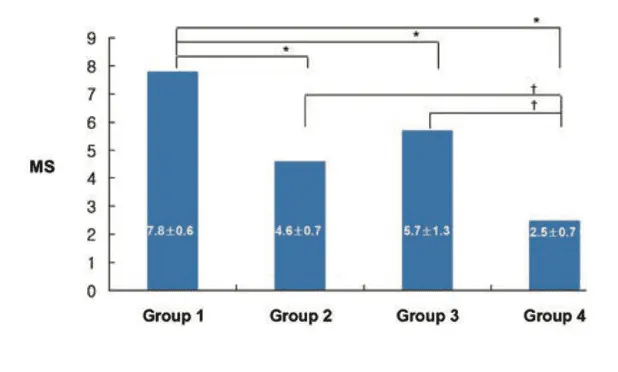
Figure 2: Changes in the histolopathological scores amongst the four experimental treatment groups.
An interesting finding from the study was the rapid reduction in lameness observed in rabbits receiving the combination of AOD9604 and HA, with full recovery noted in approximately 11 days. This suggests that the treatment may have provided not only regenerative effects but also early anti-inflammatory or analgesic benefits. Such effects could be related to pain-modulating properties observed with GH in other studies. The use of ultrasound-guided injections in the study ensured precise delivery of the treatment into the small rabbit knee joint, improving the accuracy and consistency of the intervention. Although ultrasound-guided injections are more commonly used in human clinical settings, their application in this animal study reflects an advancement in experimental methodology [1].
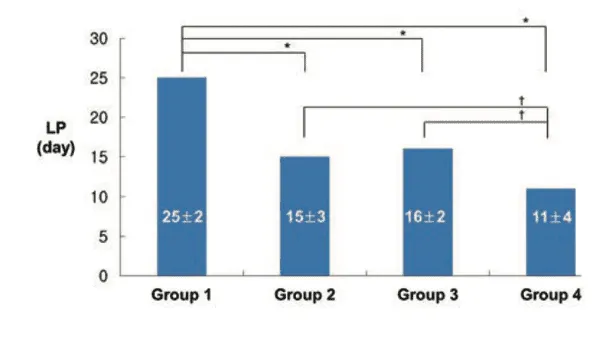
Figure 3: Changes in the lameness period amongst the four experimental treatment groups.
In conclusion, this study demonstrated that intra-articular injections of AOD9604 improved cartilage health and functional recovery in a rabbit OA model. The combination of AOD9604 with HA was more effective than either treatment alone, suggesting a synergistic interaction. Given its regenerative properties and favorable safety profile, AOD9604 holds promise as a novel therapeutic agent for OA, especially when used in conjunction with established treatments like HA [1].
2) This study performed by researchers Heffernan et al explored the chronic and acute metabolic effects of AOD9604 in several mouse models, including lean C57BL/6J, ob/ob, WT, and β3-KO mice. The primary aim was to determine AOD9604’s impact on body weight, adipose tissue mass, gene expression of β3-AR, and metabolic activity, as well as to evaluate whether the β3-AR is required for its effects [2].
In lean C57BL/6J mice, chronic treatment with hGH led to a significant increase in body weight, becoming apparent by day 8. AOD9604 caused only a modest weight increase, which was statistically significant only on the last day of the 14-day treatment period. In contrast, in the obese ob/ob mice, both AOD9604 and hGH caused a pronounced decrease in body weight. Importantly, these body weight changes were not attributable to differences in food intake across the groups, as all treatments showed similar food consumption patterns in both lean and obese mice.
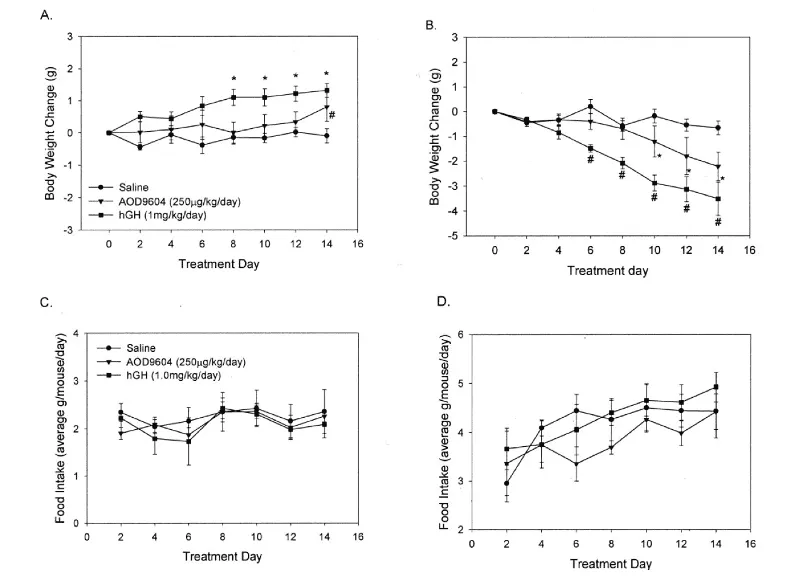
Figure 4: Changes in body weight in A) lean mice and B) obese mice and changes in food intake in C) lean mice and D) obese mice, across the three different experimental treatment groups.
Further analysis revealed differential effects on fat depots. In lean mice, neither hGH nor AOD9604 significantly altered the weight of epididymal white adipose tissue. However, in ob/ob mice, AOD9604 and hGH significantly reduced epididymal fat mass by 28% and 40%, respectively. Both compounds also decreased the mass of brown adipose tissue in lean and obese mice, suggesting a broader effect on adipose tissue metabolism [2].
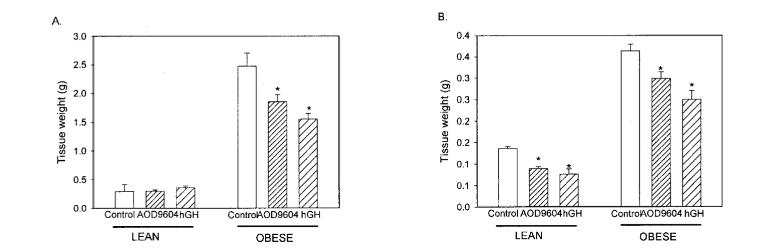
Figure 5: Changes in tissue weight in lean mice and obese mice across the three different experimental treatment groups.
To understand the underlying molecular mechanisms, the expression levels of β3-AR mRNA in both white and brown adipose tissue were assessed using reverse transcription followed by PCR and Southern blotting. Obese mice exhibited lower baseline expression of β3-AR in both fat types compared to lean controls. AOD9604 and hGH significantly upregulated β3-AR expression in the WAT of ob/ob mice, correlating with the observed reductions in adipose tissue mass. In BAT, both lean and obese mice showed increased β3-AR expression following treatment, again paralleling reductions in BAT weight.
To assess the role of β3-AR in mediating these effects, researchers examined AOD9604 and hGH treatment in β3-KO mice. Over a 28-day period, lean WT mice treated with either compound exhibited increased body weight, replicating results seen in the shorter 14-day study. However, β3-KO mice failed to show any significant changes in body weight following treatment with AOD9604 or hGH. These results indicate that β3-AR is required for the anabolic effects of these treatments on body weight [2].
When evaluating adipose tissue in WT and β3-KO mice, researchers found that neither treatment significantly affected white adipose tissue mass in either genotype. However, brown adipose tissue mass decreased significantly in WT mice treated with AOD9604 by 20% and hGH by 31%. In β3-KO mice, these reductions were absent, reinforcing the idea that β3-AR plays a necessary role in regulating the response of brown adipose tissue to these agents [2].
To further examine metabolic responses, plasma glycerol concentrations were measured as a marker of lipolysis. In WT mice, both AOD9604 and hGH elevated plasma glycerol levels, indicating enhanced lipolytic activity. This effect was significantly blunted in β3-KO mice. While hGH did produce a slight increase in glycerol in the knockout mice, the response was markedly lower than in WT animals. AOD9604 had no significant effect in the β3-KO mice. These data suggest that β3-AR is crucial for mediating the full lipolytic effects of both compounds, particularly AOD9604.
Collectively, the results of this study illustrate that AOD9604 exerts distinct metabolic effects depending on the genetic background of the animal and the presence of functional β3-ARs. In WT and obese animals, AOD9604 reduces fat mass, increases β3-AR expression, enhances lipolysis, and stimulates energy expenditure and fat oxidation. These effects are diminished or lost in β3-KO mice, indicating that β3-AR signaling is essential for many, but not all, of AOD9604’s actions. Unlike hGH, which carries broader systemic effects and risks associated with elevated IGF-1 levels, AOD9604 appears to selectively promote fat metabolism without triggering growth-related side effects, suggesting its potential as a safer therapeutic candidate for obesity and metabolic disorders [2].
Disclaimer
**LAB USE ONLY**
*This information is for educational purposes only and does not constitute medical advice. THE PRODUCTS DESCRIBED HEREIN ARE FOR RESEARCH USE ONLY. All clinical research must be conducted with oversight from the appropriate Institutional Review Board (IRB). All preclinical research must be conducted with oversight from the appropriate Institutional Animal Care and Use Committee (IACUC) following the guidelines of the Animal Welfare Act (AWA).
Citations
[1] Kwon DR, Park GY. Effect of Intra-articular Injection of AOD9604 with or without Hyaluronic Acid in Rabbit Osteoarthritis Model. Ann Clin Lab Sci. 2015;45(4):426-432.
[2] Heffernan, M., Summers, R. J., Thorburn, A., Ogru, E., Gianello, R., Jiang, W. J., & Ng, F. M. (2001). The effects of human GH and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice. Endocrinology, 142(12), 5182–5189. https://doi-org.proxy.westernu.edu/10.1210/endo.142.12.8522
AOD9604 is a peptide that is considered to be a modified fragment of growth hormone that has been shown to reduce body fat and regulate metabolism. In a study conducted by Ng et. Al, obese Zucker rats were received a dose of 500 micrograms/kg of body weight over the course of 19 days. After the 19 days, researchers discovered that compared to the control group, the experimental group of rats lost 50% body weight. Additionally it was noted that in the rats treated with AOD9604, there were increased rates of lipolytic activity, as well as no negative effect on insulin sensitivity which has been seen in animals treated with human growth hormone (https://pubmed.ncbi.nlm.nih.gov/11146367/).
A similar study conducted by Heffernan et. Al tested the effects of AOD9604 on not only lipolytic sensitivity, but also explored the pathway through which AOD9604 drives weight loss. The researchers hypothesized that weight loss with AOD9604 was driven by the beta-adrenergic pathway, more specifically the beta(3)-adrenergic receptors.
It was found through preliminary research that treatment with AOD9604 over the course of 14 days in obese mice led to decreased body weight and body fat and was shown to increase the activity of the beta(3)-adrenergic receptors in the fat cells to the same level as those in lean mice. In the next step of the experiment, the researchers removed the beta(3)-adrenergic receptors in order to see the resulting effect on weight loss. In the knockout mice, there was no significant decrease in body weight or lipolysis, thus indicating the importance of the beta-adrenergic pathways in weight loss. However, additional data was gathered with the knockout mice that indicated that while AOD9604 did not lead to significant weight loss or increase in lipolysis without the beta(3)-adrenergic receptors, there was an increase in energy expenditure and fat oxidation. Overall the study concluded that AOD9604 was shown to be beneficial in obese mice both with and without the beta(3)-adrenergic receptors present. (https://pubmed.ncbi.nlm.nih.gov/11713213/).
Heffereman et. Al conducted another study regarding the effects of AOD9604 on weight loss and fat oxidation rates, as well as to see how AOD9604 interacted with the receptor for human growth hormone (hGH). Obese and lean mice were given either saline, AOD9604, or hGH through an osmotic pump over the course of 14 days. Variables such as body weight, calorie intake, energy expenditure, glucose, and fat oxidation, insulin, and glycerol were measured before and after treatment with AOD9604 and hGH.
It was found that both AOD9604 and hGH resulted in fat loss in association with increased fat oxidation as well as increased levels of glycerol (relating to lipolysis), However, an added benefit of AOD9604 was that it did not lead to hyperglycemia in the mice or reduced insulin sensitivity. Additionally, it was noted that AOD9604 did not compete for the hGH receptor nor did it increase cell proliferation (https://pubmed.ncbi.nlm.nih.gov/11673763/).
PEPTIDES PREFER THE COLD
Always swab the top of the vial with an alcohol wipe or rubbing alcohol before accessing.
ONLY MIX WITH STERILE BACTERIOSTATIC WATER
The purity and sterility of bacteriostatic water are essential to prevent contamination and to preserve the shelf-life of dissolved peptides.
Push the pin through the rubber stopper at a slight angle, so that you inject the bacteriostatic water toward the inside wall of the vial, not directly onto the powder.
Lyophilized peptide should be stored at -20°C (freezer), and the reconstituted peptide solution at 4°C (refrigerated). Do not freeze once reconstituted.
NEVER SHAKE A VIAL TO MIX.
Air bubbles are unfavorable to the stability of proteins.
AOD9604 is sold for laboratory research use only. Terms of sale apply. Not for human consumption, nor medical, veterinary, or household uses. Please familiarize yourself with our Terms & Conditions prior to ordering.




Valoraciones
No hay valoraciones aún.